| Home Page | Embryology | Anatomy | Innervation | Physiology | Clinic Case |
 |
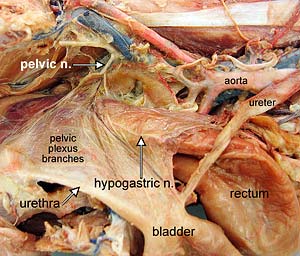 |
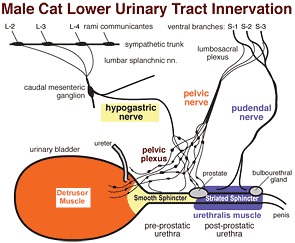
Although the ureter receives autonomic innervation, it is not functionally dependent on innervation. The urinary bladder and urethra require innervation to function effectively. They are innervated via bilateral pelvic plexuses. Part of each pelvic plexus spills onto the caudal bladder as a vesical plexus. (In contrast, the ureter can function without its autonomic innervation.)
Within pelvic plexuses, autonomic ganglia contain both sympathetic and parasympathetic postganglionic neurons, (predominantly parasympathetic). The terminal branches of postganglionic axons have preterminal and terminal varicosities (enlargements) that contain synaptic vesicles. The vesicles release neurotransmitter molecules that diffuse variable distances and bind to receptors on myocytes.
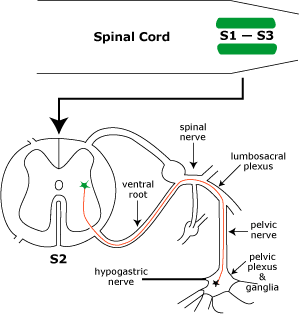
 Parasympathetic innervation to the detrusor muscle begins with preganglionic neurons located in intermediate gray matter of the sacral spinal cord (S1-3). Preganglionic axons reach the pelvic plexus via the pelvic nerve and synapse on postganglionic neurons within pelvic ganglia. Postganglionic axons release acetylcholine to cause detrusor contraction and, via nitric oxide release, internal sphincter relaxation.
Parasympathetic innervation to the detrusor muscle begins with preganglionic neurons located in intermediate gray matter of the sacral spinal cord (S1-3). Preganglionic axons reach the pelvic plexus via the pelvic nerve and synapse on postganglionic neurons within pelvic ganglia. Postganglionic axons release acetylcholine to cause detrusor contraction and, via nitric oxide release, internal sphincter relaxation.
Sympathetic innervation to the bladder and urethra originates with preganglionic neurons in the intermediolateral nucleus of the lumbar spinal cord (L2-4). The preganglionic axons travel through lumbar splanchnic nerves to the caudal mesenteric ganglion where the majority synapse. Sympathetic postganglionic axons run through the right/left hypogastric nerve and pelvic plexus to reach the bladder and urethra. The axons release norepinephrine which results in smooth muscle contraction of the bladder neck and urethra plus inhibition of the detrusor.
 |
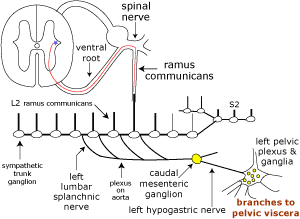 |
Somatic innervation of the urethralis muscle (as well as striated muscles of the anal canal and root of the penis) involves motor neurons in nuclei of the ventral horn of the sacral spinal cord (S2 & S3). Their axons travel through the lumbosacral plexus and pudendal nerve to release acetylcholine at neuromuscular junctions. Somatic Afferent activity from these muscles and from genitalia, anal canal, and skin of the perineum is conveyed also by the pudendal nerve.
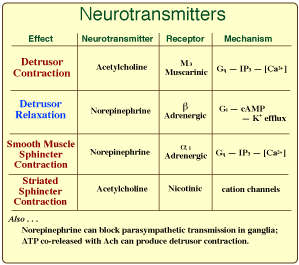 Somatic efferent neurons release acetylcholine (ACh) at neuromuscular synapses. Autonomic preganglionic neurons also release ACh at synapses with postganglionic neurons. In both cases ACh targets nicotinic ion channel receptors that increase cation permeability. The nicotinic receptors are characterized as ionotropic.
Somatic efferent neurons release acetylcholine (ACh) at neuromuscular synapses. Autonomic preganglionic neurons also release ACh at synapses with postganglionic neurons. In both cases ACh targets nicotinic ion channel receptors that increase cation permeability. The nicotinic receptors are characterized as ionotropic.
Autonomic postganglionic neurons release neurotransmitters that diffuse variable distances to bind with receptors on myocytes, generating either excitatory or inhibitory junction potentials. Excitatory potentials lead to myocyte contraction. Inhibitory potentials produce hyperpolarization and myocyte relaxation. Myocyte receptors are generally associated with G proteins and second messengers. The receptors are characterized as metabotropic.
Visceral Afferent innervation starts with bladder free nerve endings. Pain sensation is initiated by afferent axon endings in the submucosa. The axons travel through hypogastric nerves to the lumbar spinal cord. The inceptive afferent axons synapse on dorsal horn projection neurons that project their axons through the spinothalamic tract. Referred pain is the result, since somatic and visceral afferent axons synapse on the same projection neurons.
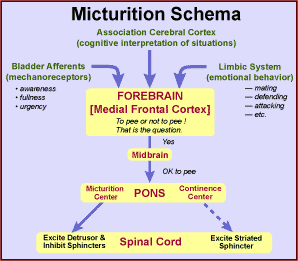
The sense of bladder fullness that leads to micturition is conveyed by axons associated with slowly adapting mechanoreceptors within the muscle coat. The axons travel through the pelvic nerve to the sacral spinal cord where they synapse on projection neurons that send axons ventrally in the lateral funiculus to the midbrain, hypothalamus and thalamus.
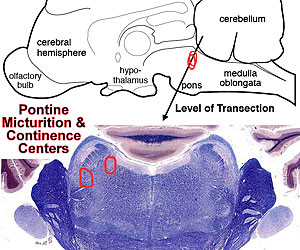
Several brain regions are concerned with micturition: A pontine micturition center enables micturition by dispatching descending axons through the lateral funiculus. The descending axons excite neurons supplying the detrusor and inhibit internal sphincter neurons. More laterally in the pons, a pontine continence center gives rise to descending axons that activate striated urethral sphincter and pelvic diaphragm muscles.
A region of the midbrain (periaqueductal gray matter) has a chief role in micturition, receiving spinal input and forebrain input and sending output axons to the pontine micturition center. The limbic system, including the hypothalamus, septum, amygdala, and cingulate cortex becomes involved because micturition timing is influenced by emotional status. Finally, the medial prefrontal cortex ultimately decides whether to urinate or not and so notifies the pons via the midbrain.
 |
 |
